Sodium stearate
« Back to Glossary Index
« Back to Glossary Index
Sodium stearate is a widely used ingredient in cosmetics, personal care, and skincare formulations. Depending on its function, it may serve as a moisturizer, preservative, emulsifier, or active ingredient to enhance the overall effectiveness and performance of a product.
Sodium stearate (Wikipedia)
Sodium stearate (IUPAC: Sodium Octadecanoate) is the sodium salt of stearic acid. This white solid is the most common soap. It is found in many types of solid deodorants, rubbers, latex paints, and inks. It is also a component of some food additives and food flavorings.
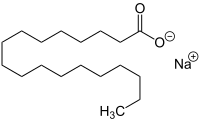 | |
| Names | |
|---|---|
| Preferred IUPAC name
Sodium octadecanoate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.011.354 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C18H35NaO2 | |
| Molar mass | 306.466 g·mol−1 |
| Appearance | white solid |
| Odor | slight, tallow-like odor |
| Density | 1.02 g/cm3 |
| Melting point | 245 to 255 °C (473 to 491 °F; 518 to 528 K) |
| soluble | |
| Solubility | slightly soluble in ethanediol |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 176 °C (349 °F; 449 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |


